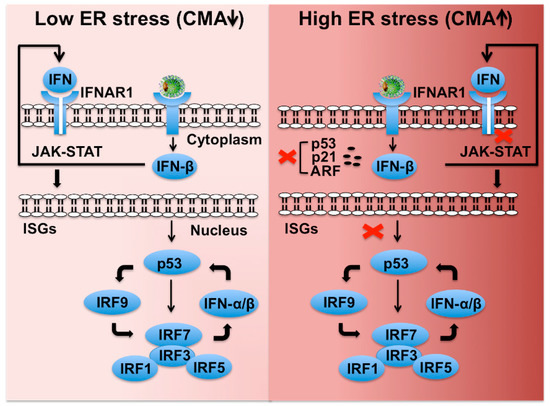
This regulation would be the molecular basis of extracellular release and propagation of misfolded proteins in various neurodegenerative diseases. To date, CMA is presumed to be restricted. Molecular chaperones in the cytosol and in the lysosomal lumen stimulate this proteolytic pathway. These findings indicate that CMA/mA and exosome release are reciprocally regulated. Chaperone-Mediated Autophagy (CMA) is a selective mechanism for the degradation of soluble proteins in lysosomes. Chaperone-mediated autophagy (CMA) is a lysosomal pathway of proteolysis that is responsible for the degradation of 30 of cytosolic proteins under conditions of prolonged nutrient deprivation. Inversely, activity of CMA/mA was significantly increased by the prevention of exosome release, which was induced by siRNA-mediated knockdown of Rab27a. Exosome release was significantly increased by the siRNA-mediated knockdown of LAMP2A, which is crucial for CMA. Although lysosomal inhibitors, NH 4Cl and bafilomycin A1, significantly increased exosome release, a MA inhibitor, 3-methyladenine, did not affect. Activation of MA and mA by rapamycin and activation of CMA by mycophenolic acid significantly decreased exosome release. In the present study, we investigated how chemical and small interfering RNA (siRNA)-mediated activation and inhibition of these autophagic pathways affect exosome release in AD293 cells. Both autophagy-lysosome proteolysis and exosome release are strongly associated with membrane trafficking. Autophagy-lysosome proteolysis is involved in protein quality control and classified into macroautophagy (MA), microautophagy (mA) and chaperone-mediated autophagy (CMA), by the routes of substrate delivery to lysosomes.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
